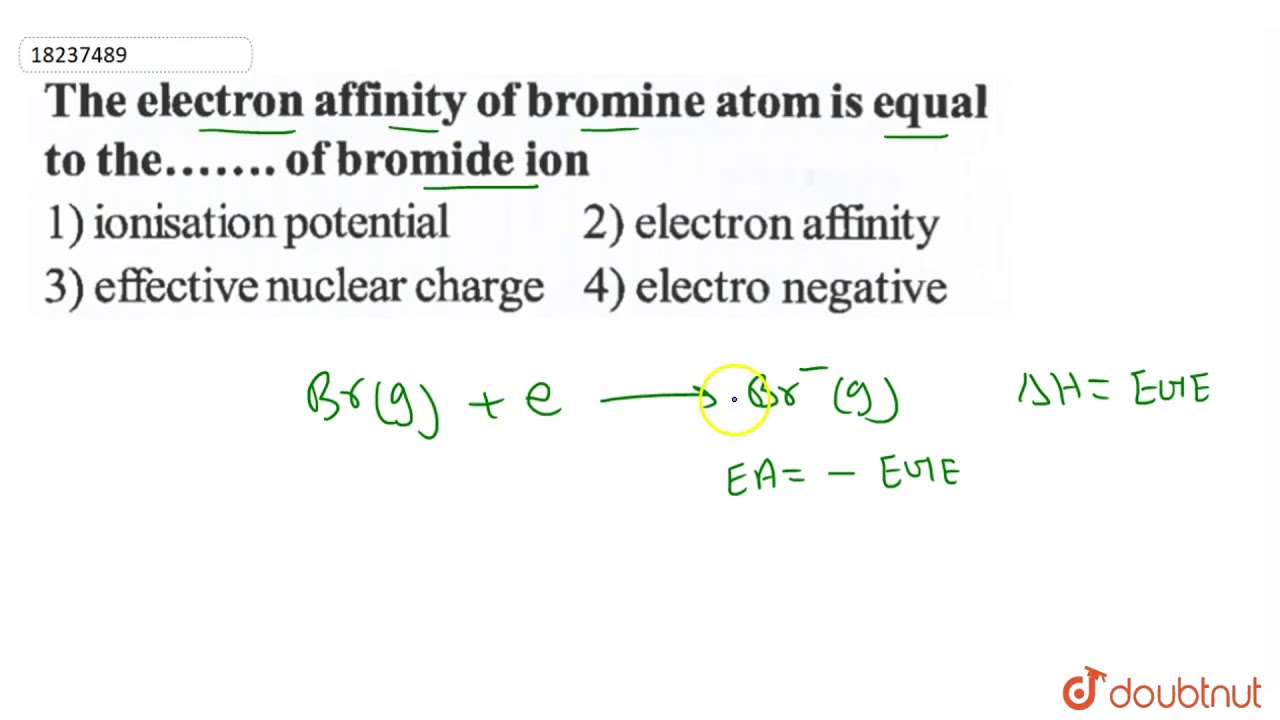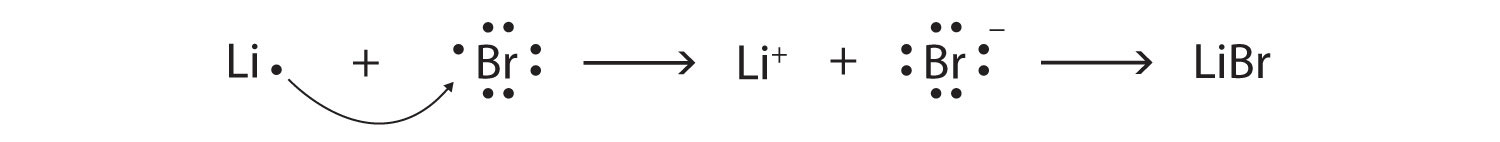SOLVED: An Ionic Bond. (a) Calculate the electric potential energy for a K^+ ion and a Br^- ion separated by a distance of 0.29 nm, the equilibrium separation in the KBr molecule.

Use Lewis symbols to represent the transfer of electrons between the following atoms to form ions with noble gas configurations: a. Ca and Br. b. K and I. | Homework.Study.com