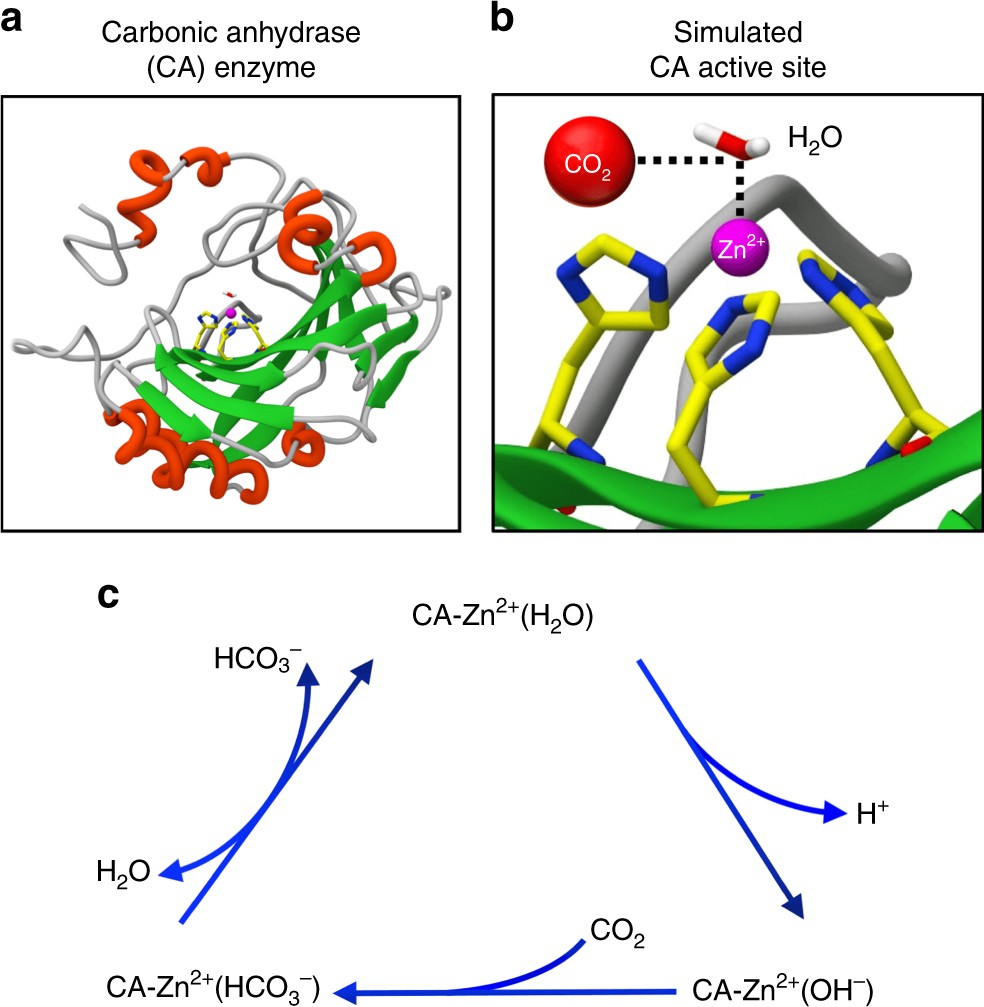
SOLVED: Calculate ΔHrxn for Ca(s) + ½ O2(g) + CO2(g) → CaCO3(s) given the following set of reactions: Ca(s) + ½ O2(g) → CaO(s) ΔHrxn = −635.1 kJ CaCO3(s) → CaO(s) +

Direct Carbonation of Ca(OH)2 Using Liquid and Supercritical CO2: Implications for Carbon-Neutral Cementation | Industrial & Engineering Chemistry Research
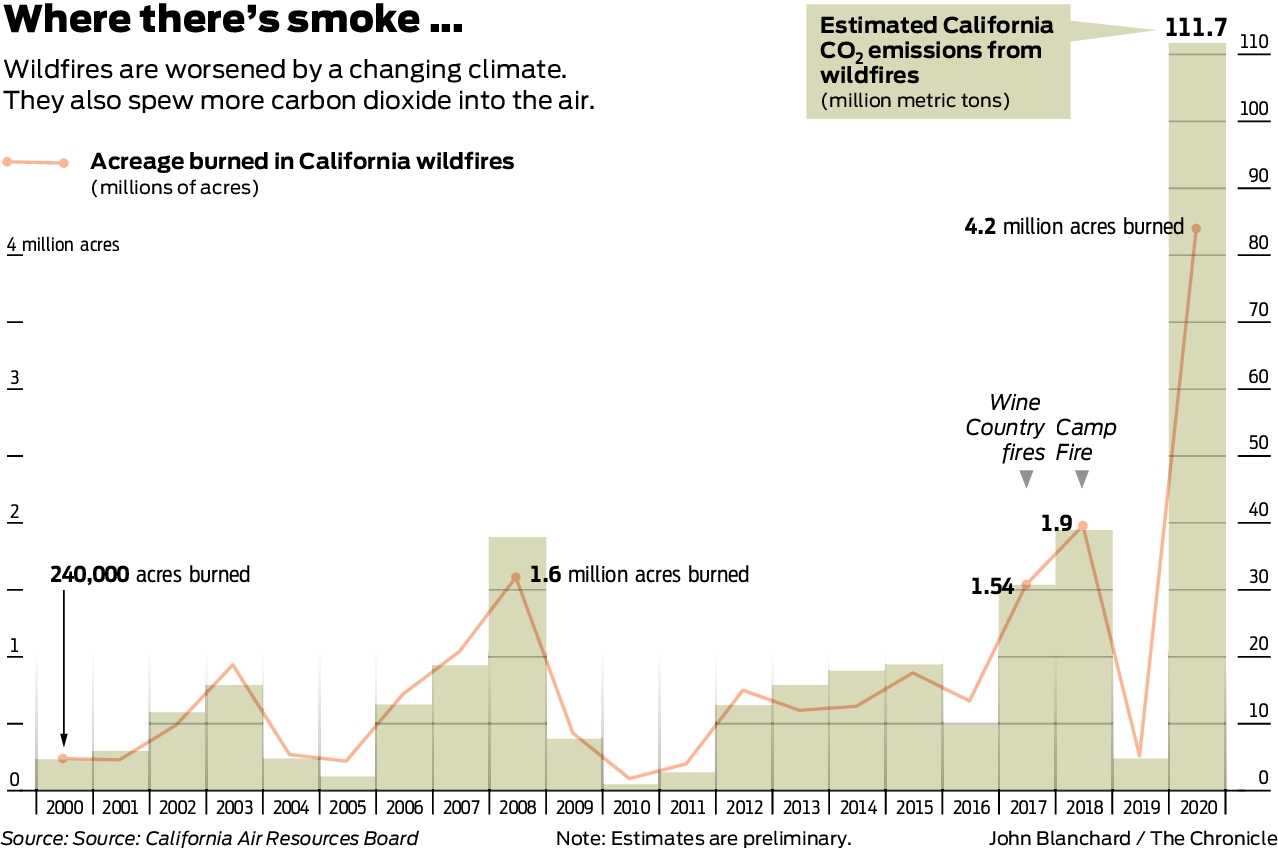
California wildfires emitted a huge amount of carbon dioxide this year. How much of a problem is that?