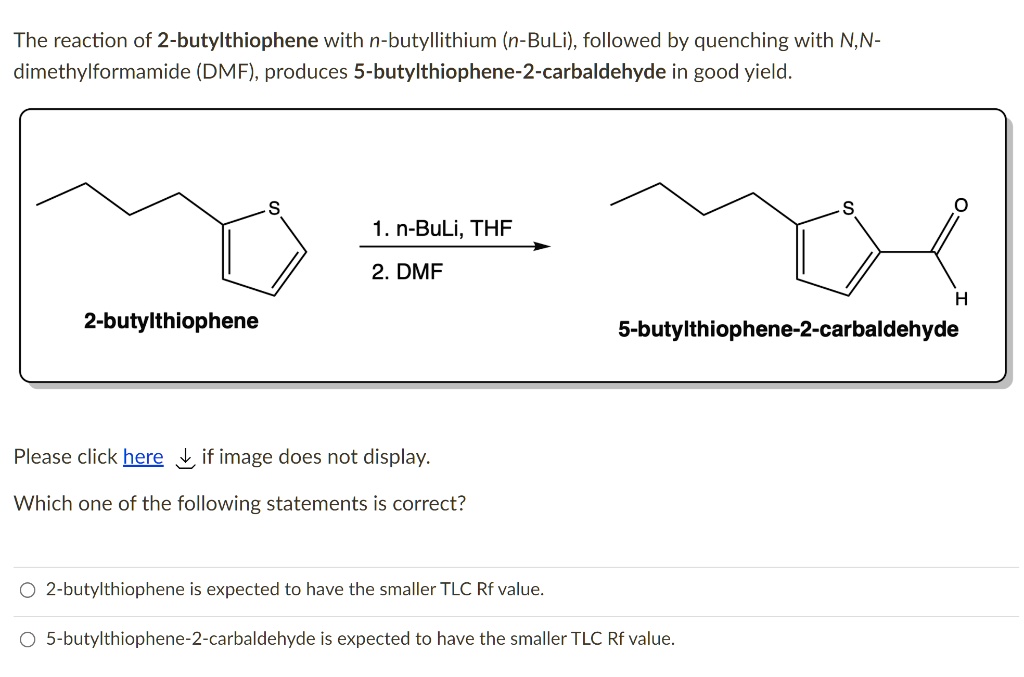
SOLVED: The reaction of 2-butylthiophene with n-butyllithium (n-BuLi), followed by quenching with N,N- dimethylformamide (DMF) , produces 5-butylthiophene-2-carbaldehyde in good yield. 1.n-BuLi; THF 2. DMF 2-butylthiophene 5-butylthiophene-2 ...

Scheme 86. Reagents: i, t-BuLi (2 equiv), Et 2 O, –78ºC; ii, TMEDA;... | Download Scientific Diagram

NMR and DFT Studies with a Doubly Labelled 15N/6Li S‐Trifluoromethyl Sulfoximine Reveal Why a Directed ortho‐Lithiation Requires an Excess of n‐ BuLi - Hédouin - Angewandte Chemie International Edition - Wiley Online Library

Halogen−Metal Exchange in 1,2-Dibromobenzene and the Possible Intermediacy of 1,2-Dilithiobenzene | The Journal of Organic Chemistry

Crystal Structures of n-BuLi Adducts with (R,R)-TMCDA and the Consequences for the Deprotonation of Benzene | Journal of the American Chemical Society
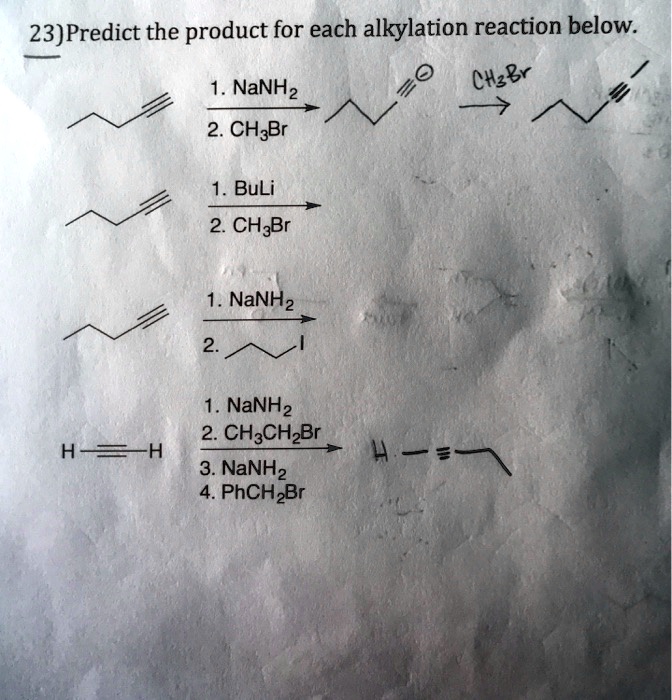
SOLVED: 23)Predict the product for each alkylation reaction below: 0 NaNH2 Chgbr 2. CHBr BuLi 2. CHzBr NaNHz NaNHz 2. CH3CHzBr 3. NaNHz PhCHzBr HS=H

![Molbank | Free Full-Text | Benzo[b]thiophene-2-carbaldehyde Molbank | Free Full-Text | Benzo[b]thiophene-2-carbaldehyde](https://www.mdpi.com/molbank/molbank-2014-M823/article_deploy/html/images/molbank-2014-M823-sch002.png)


![Solved 1n-BuLi IWWWWDK 2. EtOH Ref. (53] 0 0 \ / | Chegg.com Solved 1n-BuLi IWWWWDK 2. EtOH Ref. (53] 0 0 \ / | Chegg.com](https://d2vlcm61l7u1fs.cloudfront.net/media%2Fa30%2Fa300417d-9609-4052-99bb-061ddd14a8b5%2FphpoiZ5vq.png)